THC Oxidation to CBN
CBN is the oxidative degradation product of THC. It is more stable than THC due to its conjugated structure.
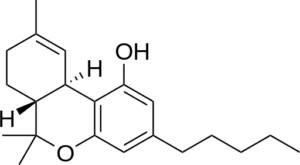
![]()
THC- Delta 9 CBN –New double bonds-Hydrogens lost here
The change is the aromatization of the top ring. 4 hydrogen atoms are lost, and two new double bonds are formed. Creating a double bond where there were hydrogens is an oxidation process, where as adding hydrogens to a double bond is a reductive process. Radical oxygen is responsible for the atmospheric degradation of THC to CBN. There are other chemical processes to synthesize CBN from THC as well.
THC Reduction to HHC

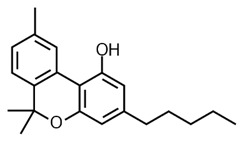
![]()
THC Delta 9 Hexahydrocannabinol (HHC)
Hexahydrocannabinol has a saturated (full hydrogen capacity, no double bonds) top ring. In this sense, it is the opposite of CBN. CBN is oxidized THC and HHC is reduced THC.
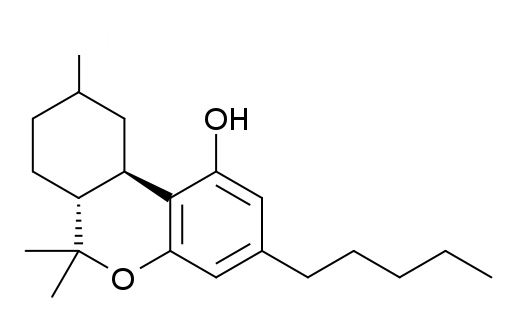
THC Delta 9 Isomerization to Delta 8 and Delta 10
THC Delta 8, Double Bond is between 8 and 9
Delta-9, the most common and the natural form of THC, is actually the least stable.
Delta 8 has the double bond in a more thermodynamically stable position, and delta 10 even more so due to its conjugation with the bottom aromatic ring.
March 24, 2019 at 3:33 pm
Dear sir
We are used lube oil recycling victory in disttiltion process.
Our problem odder smells in production
We use belching clay.
Do you have any system to remove bad odder smells for oil.
Regards
Al
Saudi Arabia
Contact 00966 555450 148
April 5, 2019 at 12:34 pm
Hello,
One of our products may suit your needs. Please contact pvt@brinstrument.com as he will be able to best help you. Thank you!
Best,
Luke
April 19, 2019 at 10:19 pm
Informative.
Only part that seems off is this “CBN is oxidized THC and HHC is reduced THC”
Is it still even Thc with a hydroxy group instead of a methyl group at the 11? ??
May 17, 2019 at 4:40 pm
Oops, that is definitely a different molecule than I meant to show. There should just be that methyl group on the top ring and not the hydroxy. Thanks!
April 19, 2019 at 10:20 pm
It would definitley be a diol of some sort..
June 20, 2020 at 4:48 pm
Sounds interesting.
December 4, 2020 at 1:09 am
[…] [7] THC isomers and reactions products, CBN, Delta 8, 9 and 10, HHChttps://brinstrument.com/blog/cannabis-distillation/thc-isomers-and-reaction-products/ […]
February 10, 2021 at 4:10 pm
I learned in Biology that delta 9 THC is the only one that breaks down in your system without leaving free radicals and that all other thc is basically giving everyone cancer.
February 11, 2021 at 7:34 pm
I don’t think that is true. At least the part about radicals.
CBN is so stable, that is how we know when ancient people used THC. And THC is physiologically safe enough that a lethal dosage is not measurable (in rats).
Now if you want to talk about side products that people are not properly removing or more exotic and unstudied isomers, then I might agree with you.
Check this out: https://en.wikipedia.org/wiki/HU-331
As with many “medicines”, dosage is key! It might even kill cancer, but that doesn’t make it good for you!
It comes back to the fact that we need to do way more research!
-Luke
October 15, 2021 at 10:44 pm
Considering that natural marijuana is one of the oldest drugs on the planet in use by humans, I’d say that if the studies haven’t been done yet that perhaps I wouldn’t trust the studies done now Considering the harmful effects of propaganda on the human mind and the human belief that studies haven’t yet been done, more likely that the studies have indeed been done but only not written in a form easily manipulated by those who do not wish for the truly therapeutic effects of marijuana. But hey believe what you want, some people believe in Jesus, or unicorns, or dragons.
February 26, 2021 at 10:26 am
Is Delta-8 thc hetzelfde als CBN?
February 26, 2021 at 5:43 pm
Is it the same as? No.
March 17, 2021 at 5:50 am
there is an effective, standard SOP for thc conversion to cbn in large scale?
March 19, 2021 at 11:58 am
Define “large”. Several Kg, yes there are people doing it. Hundred’s of Kg, no, and I don’t think there is enough demand to warrant that kind of production either.
-Luke
April 17, 2021 at 6:22 pm
Can Delta 8 be reduced to delta 9
what is the processes for hhc cbn to thc reduction? If so catalysts?
April 18, 2021 at 6:35 pm
If it could, I think it would probably be too expensive to be worth it for any kind of usage.
If you are talking about from CBD starting material, D9 is a step along the way to D8. You need to have special conditions and stop the reaction at the right time.
Those are million dollar questions. I honestly don’t know, but probably like Ni/Pd/Pt on carbon.
-Luke
July 22, 2021 at 6:29 pm
There seems to be a huge push to demonize and scare-monger about the process to create D8. The latest are claims that there are large amounts of potentially harmful stereoisomers created along with the Delta-8, and since these have not been tested, they claim a huge risk. Most recently I was shown a link from 3-Chi (producer and seller of alternative cannabinoids) that links to a Lab report from Boulder that alleges untested and illegal isomers along with the Delta-8. I am extremely skeptical of these claims, but don’t have any solid basis other than the fact that they are equating the unknown to a risk. My anecdotal experience has been that it is more stable, consistent, and medically viable than D9, and over the last year or so, there has been a huge public experiment and I have yet to hear of significant issues of any kind. I also realize that this is still anecdotal. Do you have any data to help debunk the fear?
July 26, 2021 at 4:44 pm
The whole situation legally and scientifically is very gray right now. And I will lead with that no I don’t have data.
The biggest insight I can offer is that many of these processing labs are not properly equipped for their own safety, let alone yours. I have been in many labs and seen the grass on both sides.
Similar to the black market for flower, smoking a single joint containing pesticides is not going to have long term consequences. Constant or regular exposure is an entirely different story though. I think that while over-regulation will kill the small players, some regulation is definitely needed. Has anybody died or developed serious complications in the past two years? Not that anyone can prove. But kick the can down the road 10 years of using once or more a day and we may be able to better correlate medical conditions with usage.
October 15, 2021 at 10:53 pm
Perhaps the best debunked is the question “Have there been any deaths attributed to delta 8,9,10,etc.???”. Now many current over the counter drugs are extremely dangerous, but nobody seems worried about nsaids and pop those things like tictacs. The big fear for whatever reason is “Will it get you high?” and that appears to be the long and the short of it. I enjoy it and it makes me happy, why on earth would you be so concerned about weed when alcohol is legal??? Alcohol unlike any weed product is directly attributable to the deaths of many people worldwide yet alcohol is somehow of less concern.
July 24, 2021 at 3:25 pm
Heya,
Do you think its possible to convert CBD to CBND with just heat and oxygen? (Similar to converting THC to CBN)
July 26, 2021 at 4:32 pm
Maybe.
The first problem is getting a good yield. THC + Heat Oxygen does not yield a high amount of CBN for example. Most of the lost THC goes to unwanted byproducts. The second problem is getting a proper analysis done. CBND is definitely not on the standard 10. And if you do find a lab that tests for it at all, the numbers may not be very accurate.
-Luke
August 27, 2021 at 9:08 pm
Has anyone observed what D8 dose eventually degrade into, I have heard allot of people say D8 degrades to D9. But I cant find any evidence to back that up, I think they may have just got it backwards what are your thoughts?
September 20, 2021 at 7:37 pm
D8 does not degrade to D9. D9 can be “degraded” into D8. D8 can degrade into CBN among other THC isomers.
-Luke
March 5, 2022 at 2:26 pm
What can D8 be converted to and how is Thc P Thc O made ?